|
IODOBENZENE DIACETATE | ||
|
PRODUCT IDENTIFICATION |
||
| CAS NO. |
3240-34-4 |
|
| EINECS NO. |
221-808-1 | |
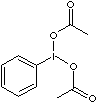
| FORMULA | C6H5I(O2CCH3)2 | |
| MOL WT. |
322.10 | |
|
H.S. CODE |
||
|
TOXICITY |
||
| SYNONYMS | Phenyliodosodiacetate; (Diacetoxyiodo)benzene; PIA; DIB; | |
| Bis(acetato)phenyliodine; Iodosobenzene diacetate; Phenyliodine(III) diacetate; Phenyliodo diacetate; Phenyliodoso acetate; Phenyliodoso diacetate; Phenyliodosyl diacetate; Bis(acetato-O)phenyliodine; 76546-99-1; 94569-95-6; | ||
| SMILES |
|
|
|
CLASSIFICATION |
|
|
|
PHYSICAL AND CHEMICAL PROPERTIES |
||
| PHYSICAL STATE |
white to off-white crystalline powder | |
|
MELTING POINT |
163 - 165 C | |
| BOILING POINT | ||
| SPECIFIC GRAVITY | ||
| pH | ||
| VAPOR DENSITY |
| |
| AUTOIGNITION | ||
|
REFRACTIVE INDEX |
||
| NFPA RATINGS | ||
| FLASH POINT | ||
| STABILITY | Stable under ordinary conditions. Light sensitive | |
|
APPLICATIONS |
||
|
Iodosobenzene diacetate is an organic reagent used as an oxidizing agent. This is suitable for cleaving glycols and alpha-hydroxy ketones in ring opening and Mannich reaction. Periodinane (hypervalent iodine) is suited for the many oxidation and rearrangements reactions including belows to prepare complex substances. Hypervalent iodine reagents are versatile synthetic tools which have low toxicity, enviromentally-friendly, mild reactivity with good yields, and ease of handling. Some common hypervalent iodines are Di(acetoxyiodo)benzene [DIB], (Bis(trifluroacetoxy)iodo)benzene [BTI], Iodosylbenzene [IOB], ((Hydroxy)(tosyloxy)iodo}benzene [HTI], 2-Iodoxybenzoic acid [IBX], Dess-Martin Periodinane [DMP].
|
||
| SALES SPECIFICATION | ||
|
APPEARANCE |
white to off-white crystalline powder | |
| PURITY | 98.0% min | |
|
MELTING POINT |
163 - 165 C | |
| TRANSPORTATION | ||
| PACKING | ||
| HAZARD CLASS | ||
| UN NO. | ||
| OTHER INFORMATION | ||
| Hazard Symbols: , Risk Phrases: , Safety Phrases: 22-24/25 | ||